Proteins are the molecular machines that power life as we know it. The rapidly-growing protein data bank now holds more than 150,000 protein structures. However, to fully understand the operation of proteins as machines, one must understand their triggered functional dynamics–how protein structures evolve in time after being triggered by an external event such as ligand docking, movement of a neighboring protein, or a change in voltage or light intensity.
Photoresponsive proteins are a particularly exciting class of proteins that, after absorbing a photon, generate mechanical energy to modulate biochemical processes and cellular behavior. This project is currently focused on the time-resolved conformational changes triggered by a pulse of light in three photoresponsive proteins: the microbial rhodopsin proton pump Proteorhodopsin, and two optogenetic proteins, a Light, Oxygen, and Voltage (LOV) sensing protein, and a cryptochrome. Proteorhodopsin belongs to the large class of 7TM proteins that make up 40% of all drug targets. LOV proteins and cryptochromes have potential as optogenetic tools — without understanding their dynamics, we cannot possibly hope to harness their motions.
The measurements are enabled by a unique methodology, under development over the last decade by the Profs. Sherwin and Han, which combines electron paramagnetic resonance (EPR) at very high magnetic fields (8.6 T) and frequencies (240 GHz) with site-directed mutagenesis and spin labeling using compounds based on Gd (III) paramagnetic metal centers.
More specifically, current goals of our project are: 1) enable our EPR spectrometer for high signal-to-noise (SNR) fast (10 microsecond repetition time) continuous wave (cw) interspin distance measurements 2) `film’ protein motion in real time by observing time-resolved lineshape changes due to dipolar-coupled spin-labelled protein residue pairs.
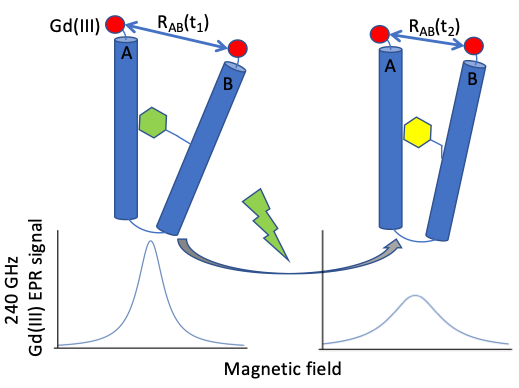
Figure 1: Cartoon depiction of EPR lineshape dependence on doubly-labelled interspin distance
In order to achieve our goal, we will take advantage of Gd-TPATCN spin labels fabricated by Dr. David Parker’s group at Durham University. Spin-7/2 Gd(III) allows for structural studies under biologically relevant conditions and interspin separations that are difficult to probe with conventional nitroxide labels. The paramagnetic core of the Gd3+ ion is well shielded by outer electrons, making it much less sensitive to the local chemical environment and will survive for days in vitro. Further, the unique chemical properties of Gd(III) permit significant resolution and distance sensitivity increases (1 nm — 5 nm) compared to cwEPR with nitroxides (1 nm — 2 nm). Gd(III) also becomes more favorable (narrow) at higher magnetic fields, which is where ITST does its EPR (8.57 T, 240 GHz).
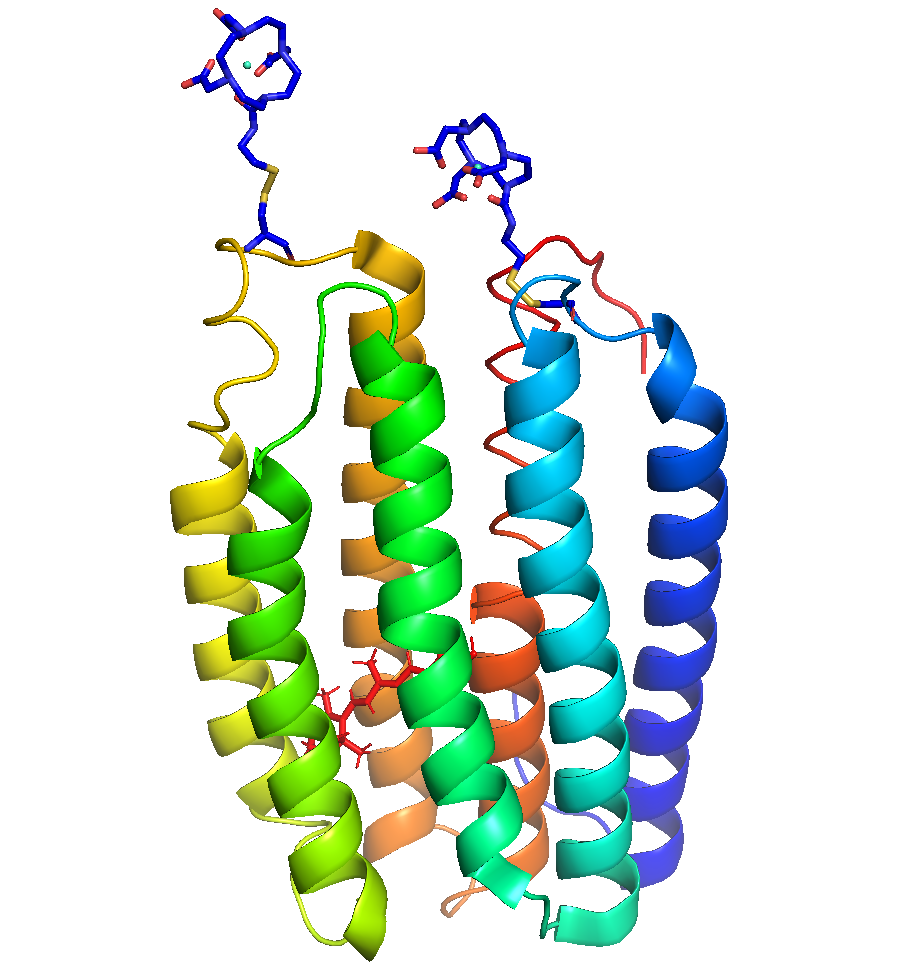
Figure 2: Spin labeling. SDSL is accomplished by introducing an EPR-active side chain at selected locations in proteins. For example, by attaching a Gd3+ based spin label on residues of interest in proteorhodhopsin.
Currently, we are interested in studying dynamic conformational changes in proteorhodopsin (PR), a green-light activated membrane-bound proton pump, and AsLOV2, a blue-light activated phototropin.
Principal Investigators:
Prof. Mark Sherwin (Physics)
Prof. Songi Han (Chemistry and Biochemistry)
Graduate Students:
Brad Price (Physics)
Shiny Maity (Chemistry)
Prof. Daniella Goldfarb, Weizmann Institute of Science
Prof. Janet Lovett, University of St. Andrews
Prof. Alex Smirnov, North Carolina State University
Prof. David Parker, Durham University
Quantitative analysis of zero-field splitting parameter distributions in Gd(III) complexes
Clayton, Jessica A. ; Keller, Katharina; Qi, Mian; Wegner, Julia; Koch, Vanessa; Hintz, Henrik; Godt, Adelheid; Han, Songi; Jeschke, Gunnar; Sherwin, Mark S. and Yulikov, Maxim
Physical Chemistry Chemical Physics 20, 10470 (2018) [www]
Gd3+-Gd3+ Distances Exceeding 3nm Determined By Very High Frequency Continuous Wave Electron Paramagnetic Resonance
Clayton, Jessica A.; Qi, Mian; Godt, Adelheid; Goldfarb, Daniella; Han, Songi; Sherwin, Mark S.
Physical Chemistry Chemical Physics 19, 5127 (2017) [www]
Determining The Oligomeric Structure Of Proteorhodopsin By Gd3+-Based Pulsed Dipolar Spectroscopy Of Multiple Distances
Edwards, Devin T.; Huber, Thomas; Hussain, Sunyia; Stone, Katherine M.; Kinnebrew, Maia; Kaminker, Ilia; Matalon, Erez; Sherwin, Mark S.; Goldfarb, Daniella; Han, Songi
Structure, 22, 1677 (2014) [www]
Extending the distance range accessed with continuous wave EPR with Gd3+ spin probes at high magnetic fields
Edwards, Devin T.; Ma, Zhidong; Meade, Thomas J.; Goldfarb, Daniella; Han, Songi; Sherwin, Mark S.
Physical Chemistry Chemical Physics, 15, 11313-11326 (2013) [www]